 AXA 37.950 € (+0,13 %)
AXA 37.950 € (+0,13 %)  BE SEMICONDUCTOR 174.850 € (-5,69 %)
BE SEMICONDUCTOR 174.850 € (-5,69 %)  EO2 2.940 € (+5,00 %)
EO2 2.940 € (+5,00 %)  AMOEBA 0.805 € (+0,63 %)
AMOEBA 0.805 € (+0,63 %)  DERICHEBOURG 7.930 € (-2,04 %)
DERICHEBOURG 7.930 € (-2,04 %)  CARREFOUR 15.335 € (-1,38 %)
CARREFOUR 15.335 € (-1,38 %)  THEON INTERNAT 28.500 € (-1,21 %)
THEON INTERNAT 28.500 € (-1,21 %)  ALTEN 52.250 € (+0,19 %)
ALTEN 52.250 € (+0,19 %)  PHARMING GROUP 1.423 € (+0,07 %)
PHARMING GROUP 1.423 € (+0,07 %)  ASM INTERNATIONAL 650.600 € (-4,30 %)
ASM INTERNATIONAL 650.600 € (-4,30 %)  BUREAU VERITAS 26.100 € (-1,51 %)
BUREAU VERITAS 26.100 € (-1,51 %)  VIVENDI SE 1.690 € (+0,66 %)
VIVENDI SE 1.690 € (+0,66 %)  UNILEVER 51.950 € (-0,44 %)
UNILEVER 51.950 € (-0,44 %)  BIOMERIEUX 89.400 € (-0,28 %)
BIOMERIEUX 89.400 € (-0,28 %)  ING GROEP N.V. 21.720 € (-0,96 %)
ING GROEP N.V. 21.720 € (-0,96 %)  REALITES 0.820 € (-8,89 %)
REALITES 0.820 € (-8,89 %)  PLANISWARE 13.960 € (-2,92 %)
PLANISWARE 13.960 € (-2,92 %)  WOLTERS KLUWER 62.320 € (-2,56 %)
WOLTERS KLUWER 62.320 € (-2,56 %)  SOLUTIONS 30 SE 0.700 € (-0,07 %)
SOLUTIONS 30 SE 0.700 € (-0,07 %)  CORBION 18.610 € (-3,02 %)
CORBION 18.610 € (-3,02 %)  BNP PARIBAS ACT.A 81.080 € (-0,96 %)
BNP PARIBAS ACT.A 81.080 € (-0,96 %)  AIR LIQUIDE 172.940 € (+0,95 %)
AIR LIQUIDE 172.940 € (+0,95 %)  HEINEKEN 65.160 € (-1,36 %)
HEINEKEN 65.160 € (-1,36 %)  IMCD 85.000 € (+2,21 %)
IMCD 85.000 € (+2,21 %)  PARROT 9.560 € (-0,83 %)
PARROT 9.560 € (-0,83 %)  PERNOD RICARD 64.700 € (+7,94 %)
PERNOD RICARD 64.700 € (+7,94 %)  MAUREL ET PROM 10.180 € (-0,39 %)
MAUREL ET PROM 10.180 € (-0,39 %)  GENFIT 8.315 € (-2,18 %)
GENFIT 8.315 € (-2,18 %)  ACCOR 40.430 € (-0,93 %)
ACCOR 40.430 € (-0,93 %)  SCHNEIDER ELECTRIC 229.200 € (-3,78 %)
SCHNEIDER ELECTRIC 229.200 € (-3,78 %)  IPSOS 33.680 € (-0,71 %)
IPSOS 33.680 € (-0,71 %)  HAFFNER ENERGY 0.057 € (-7,75 %)
HAFFNER ENERGY 0.057 € (-7,75 %)  SHELL PLC 40.150 € (-0,02 %)
SHELL PLC 40.150 € (-0,02 %)  GALAPAGOS 25.360 € (-4,01 %)
GALAPAGOS 25.360 € (-4,01 %)  MAISONS DU MONDE 0.800 € (-4,19 %)
MAISONS DU MONDE 0.800 € (-4,19 %)  VALLOUREC 21.210 € (-0,84 %)
VALLOUREC 21.210 € (-0,84 %)  FORVIA 9.618 € (+0,06 %)
FORVIA 9.618 € (+0,06 %)  2CRSI 24.600 € (+10,07 %)
2CRSI 24.600 € (+10,07 %)  TOTALENERGIES 78.490 € (-0,76 %)
TOTALENERGIES 78.490 € (-0,76 %)  BAM GROEP KON 8.445 € (-4,74 %)
BAM GROEP KON 8.445 € (-4,74 %)  ABIVAX 89.200 € (-5,91 %)
ABIVAX 89.200 € (-5,91 %)  DBT 0.044 € (-3,97 %)
DBT 0.044 € (-3,97 %)  L'OREAL 350.000 € (-0,24 %)
L'OREAL 350.000 € (-0,24 %)  AALBERTS NV 29.920 € (-0,93 %)
AALBERTS NV 29.920 € (-0,93 %)  S.E.B. 43.920 € (-0,86 %)
S.E.B. 43.920 € (-0,86 %)  AHOLD DEL 39.790 € (-0,87 %)
AHOLD DEL 39.790 € (-0,87 %)  BOLLORE 4.830 € (+0,63 %)
BOLLORE 4.830 € (+0,63 %)  ARCELORMITTAL SA 43.510 € (-2,20 %)
ARCELORMITTAL SA 43.510 € (-2,20 %)  SPIE 42.560 € (-1,98 %)
SPIE 42.560 € (-1,98 %)  ELIS 24.120 € (-0,74 %)
ELIS 24.120 € (-0,74 %)  SOCIETE GENERALE 62.280 € (-1,74 %)
SOCIETE GENERALE 62.280 € (-1,74 %)  CAPGEMINI 96.200 € (0,00 %)
CAPGEMINI 96.200 € (0,00 %)  NN GROUP 65.840 € (-0,27 %)
NN GROUP 65.840 € (-0,27 %)  KLEPIERRE 31.040 € (-0,83 %)
KLEPIERRE 31.040 € (-0,83 %)  PHILIPS KON 23.480 € (-1,01 %)
PHILIPS KON 23.480 € (-1,01 %)  VINCI 126.100 € (-0,79 %)
VINCI 126.100 € (-0,79 %)  AIRBUS 160.420 € (-1,82 %)
AIRBUS 160.420 € (-1,82 %)  RANDSTAD NV 22.910 € (-2,80 %)
RANDSTAD NV 22.910 € (-2,80 %)  RELX 27.820 € (-0,14 %)
RELX 27.820 € (-0,14 %)  MICHELIN 29.000 € (-0,75 %)
MICHELIN 29.000 € (-0,75 %) |
29/03/2026 17:05
FAST III trial demonstrates non-inferiority of CAAS vFFR to invasive wire-based FFREQS-News: Pie Medical Imaging (PMI) / Key word(s): Miscellaneous
MAASTRICHT, Netherlands, March 29, 2026 /PRNewswire/ -- Pie Medical Imaging, a global leader in cardiac imaging analysis Solutions announces the result of the FAST III trial, presented during the Late-Breaking Clinical Trials session at the Congress of the American College of Cardiology (ACC) 2026 and published in the New England Journal of Medicine. The trial confirms non-inferiority of revascularization guided by CAAS vFFR to revascularization guided by pressure wire-based FFR.
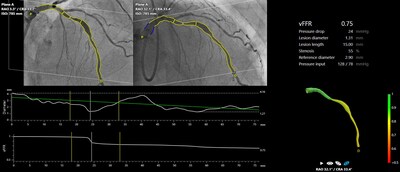
FAST III was an investigator-initiated, open label, multicenter randomized trial comparing vFFR guided versus FFR guided coronary revascularization in 2235 patients with intermediate coronary lesions (defined as 30%-80% stenosis by visual assessment or QCA). Intermediate lesions were physiologically assessed using table side vFFR or FFR and treated if vFFR or FFR ≤0.80. The primary end point was a composite of all-cause death, any myocardial infarction, or any revascularization at 1-year post-randomization. Following the tremendous efforts of the principal investigator, Dr. Joost Daemen, cardiologist in the Thoraxcenter at the Erasmus University Medical Center in Rotterdam, Netherlands. 37 participating centers and ECRI (sponsor of the trial), the trial has met its primary endpoint, showing identical MACE rates in the vFFR-guided group and the FFR-guided group. "The results of FAST III confirm the safety and feasibility of the online use of vFFR as an easy to use minimally invasive tool to guide revascularization in patients with intermediate coronary artery lesions in need of physiological lesion assessment. The technology has the potential to boost the use of physiology, which may increase the prognosis of patients in whom the decision to revascularize intermediate coronary artery lesions is still largely based on eye-balling. The use of vFFR eliminates the need for guiding catheters, invasive coronary artery instrumentation and hyperemic agents with inherent risks and patient discomfort," said Dr. Joost Daemen. Bas Kuppens, CEO at Pie Medical Imaging adds: "FAST III marks a defining moment. It confirms angiography-based physiology is non-inferior to conventional pressure wire-based physiology. Additionally, it has demonstrated benefits for care givers and patients by requiring less dose, less contrast agent, and reducing procedure lead time". Doris Pommi, head of Cardiovascular Care at Siemens Healthineers, added: "The results of the FAST III study mark a significant milestone for cardiovascular care in Europe and for our partnership with Pie Medical Imaging. Its success shows how much we can achieve when we join forces and combine scientific excellence, clinical experience, and industrial innovation. (The patient impact of the study is substantial, we are looking at safer, faster, less uncomfortable coronary assessments with potentially lower costs and wider access to physiologic lesion evaluation.)" The trial is funded by research grants from Pie Medical Imaging (Maastricht, The Netherlands) and Siemens Healthineers AG (Forchheim, Germany). The study is sponsored by ECRI (European Cardiovascular Research Institute, Rotterdam-the Netherlands). Cardialysis (Rotterdam, The Netherlands) is responsible for trial services including trial management and Core Laboratory activities. About Pie Medical Imaging Pie Medical Imaging is a global leader in advanced cardiovascular image analysis, quantification and visualization. Based in Maastricht, The Netherlands, the company oversees worldwide innovation and commercialization of the CAAS and 3mensio product lines. With decades of expertise and deep commitment to innovation, Pie Medical Imaging delivers software solutions that empower clinicians, researchers, and industry partners to enhance diagnosis, procedure planning, and image-guided interventions. The company complies with international quality and regulatory standards, ensuring high reliability and clinical confidence. Pie Medical Imaging is part of the Esaote Group, a leader in the biomedical equipment sector. More information about Pie Medical Imaging is available at www.piemedicalimaging.com.

Photo - https://mma.prnewswire.com/media/2943619/FAST_III_PR_Image.jpg
 29.03.2026 CET/CEST Dissemination of a Corporate News, transmitted by EQS News - a service of EQS Group. 2299744 29.03.2026 CET/CEST Source : Webdisclosure.com |
Cotations différées d'au moins 15 minutes (Paris, Amsterdam, Bruxelles, Lisbonne).
Cotations à la clôture (Francfort, New-York, Londres, Zurich).
Flux de cotations : Euronext (Places Euronext et Cours des Devises).
Bourse : technologie Cote Boursière
